Health
Results: 6937-6948 of 8689

Takeda Announces U.S. FDA Approval of Supplemental New Drug Application (sNDA) for ICLUSIG® (ponatinib) in Adult Patients with Newly Diagnosed Ph+ ALL

DEXIS Enhances its Dental Implant Ecosystem with Intraoral Scanning Software Update - DEXIS IS ScanFlow

Award-winning journalist, Lara Setrakian, launches 'HealthBeats' podcast in partnership with Abu Dhabi Global Healthcare Week

Moon Surgical's Maestro System powered by NVIDIA Holoscan paves the way to next-generation laparoscopy, with over 200 patients treated
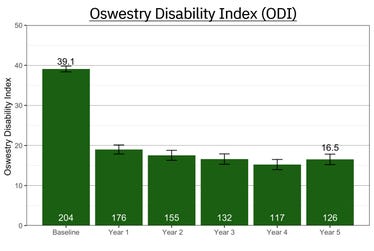
Mainstay Medical Announces Publication of the First Neuromodulation Study for Low Back Pain with 5-Year Follow-Up

Forge Biologics’ Novel AAV Gene Therapy FBX-101 for Patients with Krabbe Disease is Granted UK’s Innovation Passport Designation




